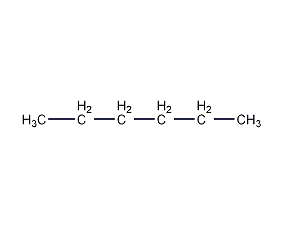
Structural formula
| Business number | 031E |
|---|---|
| Molecular formula | C6H14 |
| Molecular weight | 86.18 |
| label |
dipropyl, n-hexane, Isohexane, n-Hexane, Aliphatic hydrocarbons |
Numbering system
CAS number:110-54-3
MDL number:MFCD00009520
EINECS number:203-523-4
RTECS number:MN9275000
BRN number:1730733
PubChem number:24857835
Physical property data
1. Characteristics: Highly volatile colorless liquid with gasoline smell. [1]
2. Melting point (℃): -95.3~-94.3[2]
3. Boiling point ( ℃): 69[3]
4. Relative density (water=1): 0.66[4]
5 .Relative vapor density (air=1): 2.97[5]
6. Saturated vapor pressure (kPa): 17 (20℃)[6]
7. Heat of combustion (KJ/mol): -4159.1[7]
8. Critical temperature (℃): 234.8 [8]
9. Critical pressure (MPa): 3.09[9]
10. Octanol/water partition coefficient: 3.9 [10]
11. Flash point (℃): -22[11]
12. Ignition temperature ( ℃): 225[12]
13. Explosion upper limit (%): 7.5[13]
14. Explosion Lower limit (%): 1.1[14]
15. Solubility: Insoluble in water, soluble in most organic solvents such as ethanol, ether, acetone, and chloroform. [15]
16. Total combustion calorific value (KJ/mol): 4165.9
17. Minimum combustion calorific value (KJ/mol): 3857.6
18. Viscosity (25℃, liquid)/mPa·s: 0.307
19. Heat of evaporation (0ºC) (kJ/mol): 33.12
20. Heat of fusion (kJ/mol): 13.04
21. Aniline point (ºC): 63.6
22. Thermal conductivity (25 ºC, liquid)/[W/( m·K)]: 116.81×10-3
23. Heat of formation (25 ºC, liquid) / (kJ·mol): -198.96
24.Specific heat capacity (0 ºC, constant pressure, liquid)/[kJ/(kg·K)]: 2.278
Toxicological data
1. Acute toxicity[16]
LD50: 25g/kg (rat oral)
LC50: 48000ppm (rat inhalation, 4h)
2. Irritation[17] Rabbit eye: 10mg, mild irritation.
3. Subacute and chronic toxicity[18] Rats inhale 2.76g/m3 every day , lasted for 143 days, with decreased nighttime activity, mild abnormal reaction of the reticuloendothelial system, myelin degeneration of peripheral nerves, mild changes in axons, and mild atrophy of gastrocnemius muscle fibers.
Ecological data
1. Ecotoxicity[19]
LC50: 4mg/L (24h) (goldfish); >50mg/L (24h) (Daphnia)
IC50: 10mg/L (72h) (algae)
2. Biodegradability [20] MITI-I test, initial concentrationDegree 100ppm, sludge concentration 30ppm, 100% degradation after 4 weeks.
3. Non-biodegradability[21] In the air, when the hydroxyl radical concentration is 5.00×105 pieces/cm3, the degradation half-life is 3d (theoretical).
4. Bioconcentration[22] BCF: 200 (theory)
Molecular structure data
1. Molar refractive index: 29.84
2. Molar volume (cm3/mol): 127.5
3. Isotonic specific volume (90.2K ): 270.8
4. Surface tension (dyne/cm): 20.3
5. Dielectric constant (F/m): 1.87
6. Polar Chemical rate (10-24cm3): 11.83
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 3
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 6
8. Surface charge: 0
9. Complexity: 12
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Stability[23] Stable
2. Incompatible substances[24] Strong oxidants, strong acids, strong bases, halogens
3. Polymerization hazards[25] No polymerization
Storage method
Storage Precautions[26] Stored in a cool, ventilated warehouse. Keep away from fire and heat sources. The storage temperature should not exceed 29°C. Keep container tightly sealed. should be kept away from oxidizer, do not store together. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
1. Exists in straight-run gasoline, platinum reforming raffinate or wet natural gas, with a content of 1% to 15%. At present, industrial production mainly separates the raffinate from the platinum reforming unit (containing 11% to 13% hexane). After removing the light and heavy components by distillation, a fraction containing 60% to 80% n-hexane is obtained. Double-tower continuous distillation is used, and then hydrogenation is carried out using the 0501 catalyst to remove benzene and other unsaturated hydrocarbons to obtain qualified products. The United States also uses adsorption separation method to prepare n-hexane.
2. Use n-hexane as raw material (n-hexane content is 90% to 95%), and obtain high-purity n-hexane through precision distillation.
3. Mainly adopt the raffinate oil separation method. The raffinate oil from the platinum reforming unit (containing 11%~13% ethane) is distilled and separated. After removing the light and heavy components, n-hexane is obtained with a purity of 60%~80%, and then used as 60~ The fraction at 140°C is used as raw material (containing 11%~13% n-hexane). Two towers are used for continuous separation. After removing light and heavy components, a fraction containing 60%~80% n-hexane is obtained. Finally After hydrogenation with 0 5 0 1 catalyst, unsaturated hydrocarbons such as benzene are removed to obtain n-hexane.
Purpose
1. Mainly used as solvents, such as solvents for the polymerization of propylene and other olefins, extractants for edible vegetable oils, solvents for rubber and coatings, and diluents for pigments. Plus, it’s a high-octane fuel.
2. Mainly used for atmospheric monitoring and preparation of standard gas and calibration gas.
3. Used as solvent and reference material for chromatographic analysis. Used for the determination of refractive index, determination of moisture in methanol, and also used in organic synthesis.
4. Mainly used as solvent. It is used as a solvent in cosmetics, mainly as a solvent for cellulose in cosmetics such as nail polish.
5. Used in organic synthesis, as solvents, chemical reagents, paint thinners, media for polymerization reactions, etc. [27]

 微信扫一扫打赏
微信扫一扫打赏

