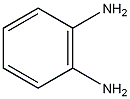
Structural formula
| Business number | 029B |
|---|---|
| Molecular formula | C6H8N2 |
| Molecular weight | 108 |
| label |
1,2-phenylenediamine, O-phenylenediamine, 1,2-diaminobenzene, OPD, Aromatic nitrogen-containing compounds and their derivatives |
Numbering system
CAS number:95-54-5
MDL number:MFCD00007721
EINECS number:202-430-6
RTECS number:SS7875000
BRN number:606074
PubChem number:24856671
Physical property data
1. Character: colorless monoclinic crystal [6]
2. pH value: 8.7 (1% solution) [7]
3. Melting point (℃): 102~104[8]
4. Boiling point (℃): 252~258[9]
5. Relative density (water=1): 1.03[10]
6. Relative vapor density (air=1): 3.7[11]
7. Saturated vapor pressure (kPa): 0.33 (100℃)[12]
8 .Critical pressure (MPa): 5.18[13]
9. Octanol/water partition coefficient: 0.15[14]
10. Flash point (℃): 156 (CC) [15]
11. Explosion limit (%): 9.8[16]
12. Lower explosion limit (%): 1.5[17]
13. Solubility: slightly soluble in cold water, easily soluble in ethanol, ether, Chloroform. [18]
Toxicological data
1. Acute toxicity[19] LD50: 1070mg/kg (rat oral)
2. Irritation No information available
3. Mutagenicity[20] Microorganisms Mutagenicity: Salmonella typhimurium 1g/L. Micronucleus test: mice take 216mg/kg orally (24h). DNA damage: human lymphocytes 15mmol/L. DNA inhibition: human HeLa cells 7mmol/L. Cytogenetic analysis: human lymphocytes 10mmol/L.
Ecological data
1. It is extremely harmful to water and toxic to fish. Do not let the product enter the water body.
2. Ecotoxicity[21] LC50: 24mg/L (96h) (zebrafish); 280mg/ L (96h) (bluegill sunfish)
3. Biodegradability No data available
4. Non-biodegradability[22] In the air, when the concentration of hydroxyl radicals is 5.00×105/cm3 When, the degradation half-life is 2.2h (theoretical).
Molecular structure data
1. Molar refractive index: 34.72
2. Molar volume (cm3/mol): 93.9
3. Isotonic specific volume (90.2K ): 258.9
4. Surface tension (dyne/cm): 57.5
5. Polarizability (10-24cm3): 13.76
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 2
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bondsRaw materials and cationic dyes, used to produce fur yellow brown M, cationic dyes, vat red GG, vat orange GR. Also used in the manufacture of polyamide, polyurethane and leveling agents. It is used in the pesticide industry to synthesize systemic fungicides (carbendazim, thiophanate and thiophanate-methyl).
2. Synthesize a variety of heterocyclic intermediates. Used to prepare antioxidants.
3. O-phenylenediamine is the main intermediate for pesticides, medicines, dyes, and developing rubber agents. O-phenylenediamine and its derivatives can be used as starting materials to react with carbonyl compounds through thermal reaction and microwave irradiation to form benzopolynitrogen heterocyclic aromatic compounds, such as benzimidazoles, quinoxalines and benzazepines. Miscellaneous categories, etc.
When o-phenylenediamine reacts with 2-pyridinecarboxaldehyde, the N4 Schiff base ligand is obtained1[1], but when 2 When -pyridine carboxaldehyde was replaced with 2-acetylpyridine, using methyl chloroformate as the catalyst, the corresponding N4 Schiff base ligand2 was not obtained in the reaction in isopropanol, but benzene was obtained. The six-membered ring product is 2-(2′-pyridyl)quinoxaline3[2].
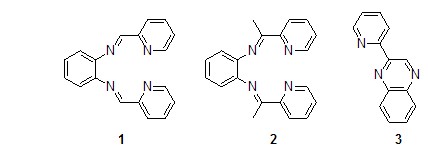
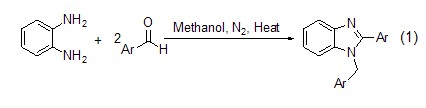
O-phenylenediamine and two equivalents When the aromatic aldehyde reacts in methanol, the corresponding five-membered nitrogen heterocyclic benzimidazole derivative (formula 1)[3] is generated.
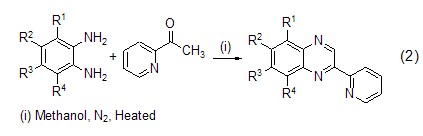
Utilizing o-phenylenediamine and its The derivative was reacted with 2-acetylpyridine as raw materials, p-toluenesulfonic acid as catalyst, in methanol solvent, heated (50~60 ºC) and stirred for 10~20 hours to obtain a benzo six-membered ring product (Formula 2) [4,5].
The raw material aromatic aldehyde in formula 1 When replaced by an acetyl aromatic compound, a benzo seven-membered ring product, that is, a benzazepine compound (Formula 3) is obtained.
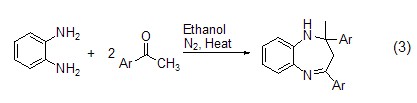
Change the reaction method to microwave radiation Benzazepine compounds (Formula 4) can also be obtained by combining o-phenylenediamine and its derivatives with acetyl aromatic compounds.
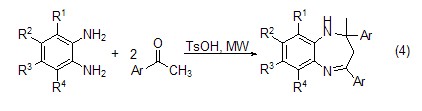
4. As a pesticide intermediate, Dye intermediates. [29]

 微信扫一扫打赏
微信扫一扫打赏

