Background and overview[1-2]
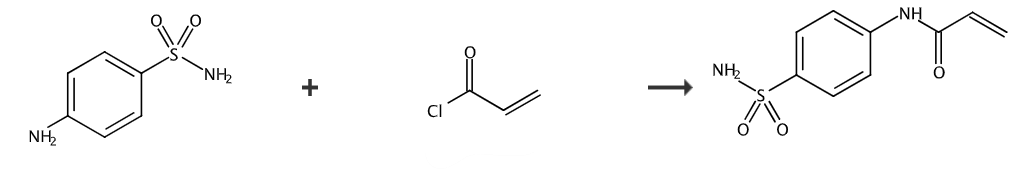
N-[4-(Sulfonamide)phenyl]acrylamide (ASPAA) monomer can be homopolymerized or copolymerized with other monomers to form pharmacologically active polymers that are water-soluble, have little toxic and side effects, and have slow Large molecule drugs that release effects. At the same time, because the sulfonamide group has a certain alkali solubility, copolymers of ASPAA and acrylic esters and other monomers can be used to prepare lithographic and computer-to-plate printing plates with good properties such as alkali developability, development latitude, and print durability.
Preparation[1][3]
Report 1,
Add sulfonamide (1.00g) and pyridine (1.41mL) to 1,4-dioxane (30mL). Acryloyl chloride (0.49 mL) was added dropwise and the solution was stirred at room temperature for 3 hours. 1M HCl was added and the solution was extracted with ethyl acetate. The extract was dried over brine and anhydrous sodium sulfate, and the solvent was removed to give a crude product that was pure enough for subsequent use.
Report 2,
Add 13.76g (80mmol) of p-aminobenzenesulfonamide, a certain amount of NaHCO3, 20mg of 1,3-dinitrobenzene, and 100mL of DMF into a dry three-necked flask. Use ice for the reaction solution Cool the bath to 0°C to 2°C, slowly add 7.96g (88mmol) of acryloyl chloride through a constant pressure dropping funnel while stirring, and react at a certain temperature for 3 hours after the dropwise addition is completed. Filter to remove the salt, and pour the filtrate into 10 times the volume of methanol-water to precipitate a white solid. Filter, wash the filter cake with water 3 to 5 times, and then recrystallize it with methanol to obtain ASPAA. The yield is 50% to 65%, m.p. 234℃~ 236℃;1HNMRδ: 10.5 (s, 1H, CONH), 7.83~7.85 (d, 2H, ArH), 7.79~7.80 (d, 2H, ArH), 7.29 (s, 2H, NH2), 5.81, 6.33 (m, 2H, =CH2), 6.47 (t, 1H, =CH); IR ν: 3342 (N-H), 3133, 3066 (=C-H), 1676 (C=O, amide I spectrum band), 1536 (CONH, amide II band), 1593, 1493 (-C=CH), 1411 (C-N stretching vibration), 1164 (SO2-N), 836 (benzene ring disubstituted) cm-1 ;Ana.lcalcd for C9H10N2O3S:C47.78, H4.45, N12.38; found C 47.12, H4.33, N12.21
References
[1]FromPCTInt.Appl., 2010065865, 10Jun2010
[2] Gao Yingxin, Bao Yongzhong, Huang Zhiming, Weng Zhixue. Reactivity rate of copolymerization of N-[4-(sulfonamide)phenyl]acrylamide with acrylonitrile and methyl methacrylate[J]. High Acta Molecule, 2004(03):450-453.
[3] Gao Yingxin, Bao Yongzhong, Huang Zhiming, Weng Zhixue. Synthesis of N-[4-(sulfonamide)phenyl](meth)acrylamide [J]. Synthetic Chemistry, 2005(01):22- 24+36-1.

 微信扫一扫打赏
微信扫一扫打赏

